

All Choked Up
It’s not uncommon in ALS for something unexpectedly and abruptly to aggravate your airway and throw you into a choking fit. I know from personal experience that these choking, gagging, gasping spells are physically exhausting, frustrating and upsetting for both the choker and the caregiver.The actions to take for choking depend on the type of choking spell you’re having. For example: Read More
Recent ALSN Articles

Tips for Tackling Depression
A bout of depression can bring added difficulty to a life already complicated by ALS. To keep depression from bringing you down, check out these 10 tips — culled from a variety of expert sources — for understanding, spotting and stopping it.1.) A common misconception about depression in people with terminal illness is that in such situations depression is “understandable” or “normal” and nothing to worry about. In fact, studies have shown that most people with late-stage ALS aren’t depressed. Identifying and treating depression, rather than accepting it as “normal,” can have a tremendous positive effect on quality of life.
Read More
Tips for Coping with Summer’s Heat
Summer typically is the time when people head out and enjoy the great outdoors. But people with ALS have to take a few extra precautions during these hot months, warns Lora L. Clawson, certified registered nurse practitioner and director of ALS clinical services at the MDA/ALS Center at Johns Hopkins University in Baltimore.
Read MoreBaby, It’s Cold Outside!
When it comes to cold weather and ALS, 53-year-old Pati Milewski, of Olympia, Wash., likens her experience to that of “a house with no thermostat.”Although inability to regulate body temperature isn’t recognized as a classic symptom of ALS, decreased mobility limits the ability to stay warm by being active.
Read More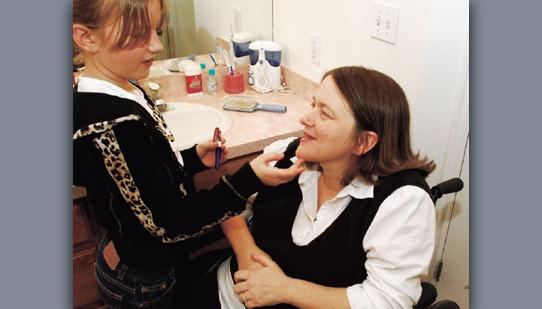
Role Reversal: When Children Care for a Parent with ALS
In Sandstone, Minn., Alea, 16, and Alexandra, 13, have added “caregiver” to the long list of roles they assume in life, including daughter, sister, friend, teenager and student. The girls’ father, William Anderson, learned he had ALS on March 26, one day after his 49th birthday.To date, Anderson’s symptoms include weakness and lack of coordination in his hands, arms, shoulders, chest and back. He retired from his job as a corrections officer with the state of Minnesota due to the progression of his symptoms, but still serves as a volunteer with, and president of, the Pine County Sheriff’s Department, planning meetings and events. He uses braces to preserve strength in his wrists, but even so has trouble managing forks and spoons, getting dressed, handling tools such as hammers and wrenches, cooking and folding clothes.
Read MoreTips for Helping Your Caregiver Cope
A tense caregiving relationship can be difficult for everyone involved.Although the focus often is on the person with ALS, sometimes caregivers also need help dealing with their feelings and learning new skills, including how to ask for help from others, says Elizabeth Etigson, a licensed professional clinical counselor who facilitates the MDA/ALS support group in Albuquerque, N.M., and has a private practice, Purple Turtle Counseling.
Read More
Water-Contamination Link to ALS Tenuous Without More Data
Recently, media outlets reported a possible link between developing ALS and living near Lake Mascoma in Western New Hampshire.The Union Leader in New Hampshire and other news outlets indicated that the risk of developing ALS is “25 times” higher than average for people living around Lake Mascoma, located in Enfield and Lebanon, N.H. The source of this statistic was not explained.
Read MoreALS Research: Risk Raisers
A large, multinational study to identify genetic risk factors associated with amyotrophic lateral sclerosis (ALS) has found two DNA sequences on chromosomes 9 and one on chromosome 19 that are significantly different in people with and without the disease and may contribute to its development.The identified DNA regions on both chromosomes contain genes for biological processes that could have an effect on the disease.
Read More
Not Gloom and Doom: Demystifying Hospice
For some, “hospice” is a vague idea whose connection to death leads to outright rejection before the true facts are known.In fact, hospice neither prolongs life nor hastens death. Instead, it offers practical care and support at a difficult and sometimes traumatic time.“People should think about hospice as a comfort measure, not gloom and doom,” says Janet Neigh, executive director of the Hospice Association of America (HAA). “There’s a misconception that hospice is a place. It’s actually a concept, an approach to care for people who have a terminal illness.”
Read More
To Cath or Not to Cath?
When ALS weakened Jane Cheng’s mother, her caregivers found it took a great deal of strength to help her transfer on and off the toilet.“I twisted my ankle a few times when attempting to turn the pivot disk with my foot while supporting most of her weight,” says Cheng, who cares for her mother in central Pennsylvania.
Read More
ALS: Not Just About Motor Neurons Anymore
Not long ago, if you asked any neurologist about ALS, you’d have been told that it’s a disease of the motor neurons — nerve cells in the brain and spinal cord that control voluntary muscle movement. That’s still true. But the last few years have added clauses to that description that recognize the important contributions of other cells to the deadly disease process. That could be good news, because those other cells may be easier targets than motor neurons.
Read MoreMDA Resource Center: We’re Here For You
Our trained specialists are here to provide one-on-one support for every part of your journey. Send a message below or call us at 1-833-ASK-MDA1 (1-833-275-6321). If you live outside the U.S., we may be able to connect you to muscular dystrophy groups in your area, but MDA services are only available in the U.S.